
In 1991, Michael J. Fox’s life was changed forever. Just months after the release of the third movie in the hugely successful Back to the Future trilogy, the actor visited a neurologist after experiencing muscle pain and was diagnosed with Parkinson’s. Fox was 30 at the time and at the peak of his career. Since then, he has lived a more or less normal life, with periods of greater difficulty than others, and only acting in a part-time capacity before deciding to definitively retire in 2020.[1]
What is Parkinson’s disease? Is it considered a severe disease? How does a person get it? Can it be cured? Parkinson’s is actually not rare, but when we talk about it, there are still countless questions lingering in our minds. Herein, we introduce Parkinson’s disease and its related mouse models used in research to better understand this long-term neurodegenerative disorder that affects 10 million people worldwide.

Figure 1. Michael J. Fox during the 'Back to the Future' panel at Comic-Con in New York on October 8, 2022. (Source: MIKE COPPOLA (GETTY IMAGES))
Parkinson's disease (PD), or simply Parkinson's, is a long-term degenerative disorder of the central nervous system (CNS) that mainly affects the motor system, first described systematically by the English doctor James Parkinson. Its clinical symptoms mainly include tremors, rigidity, slowness of movement, and difficulty with walking, and non-motor symptoms of sleep disorders, cognitive impairment, and depression.[2]
As the second most prevalent neurodegenerative disease in the world, the incidence of PD is increasing year by year, and the growth rate of disability and death caused by it is higher than that of any other neurological diseases. According to WHO statistics, as of 2019, more than 8.5 million people worldwide suffer from PD. Among them, 5.8 million people were disabled by PD, an increase of 81% compared with 2000, and 329,000 patients died, which doubled since 2000.[3]
The exact pathogenesis of PD is unknown, but both genetic and environmental factors are believed to play a role in its development. Diagnosis of typical cases is mainly based on symptoms, with motor symptoms being the chief complaint. No cure for PD is known; current treatment aims to reduce the effects of the symptoms.
The clinicopathological features of PD are the loss of neurons in the substantia nigra pars compacta (SNpc) in the specific region of the midbrain (Figure 2), Lewy bodies (LB) formed by the accumulation of misfolded α-synuclein (α-Syn) (Figure 3), as well as striatal dopamine (DA) deficiency.[5] Currently, the mainstream theories on the pathogenesis of PD include α-synuclein misfolding and aggregation, mitochondrial dysfunction, impairment of protein clearance, neuroinflammation, and oxidative stress.[4]
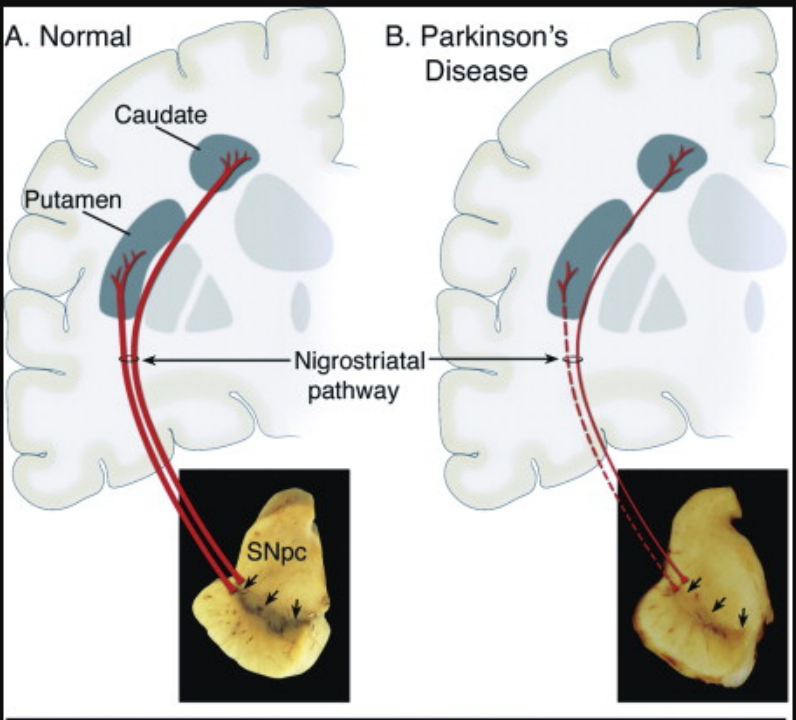
Figure 2. Neuropathology & Clinicopathological Features of Parkinson's Disease
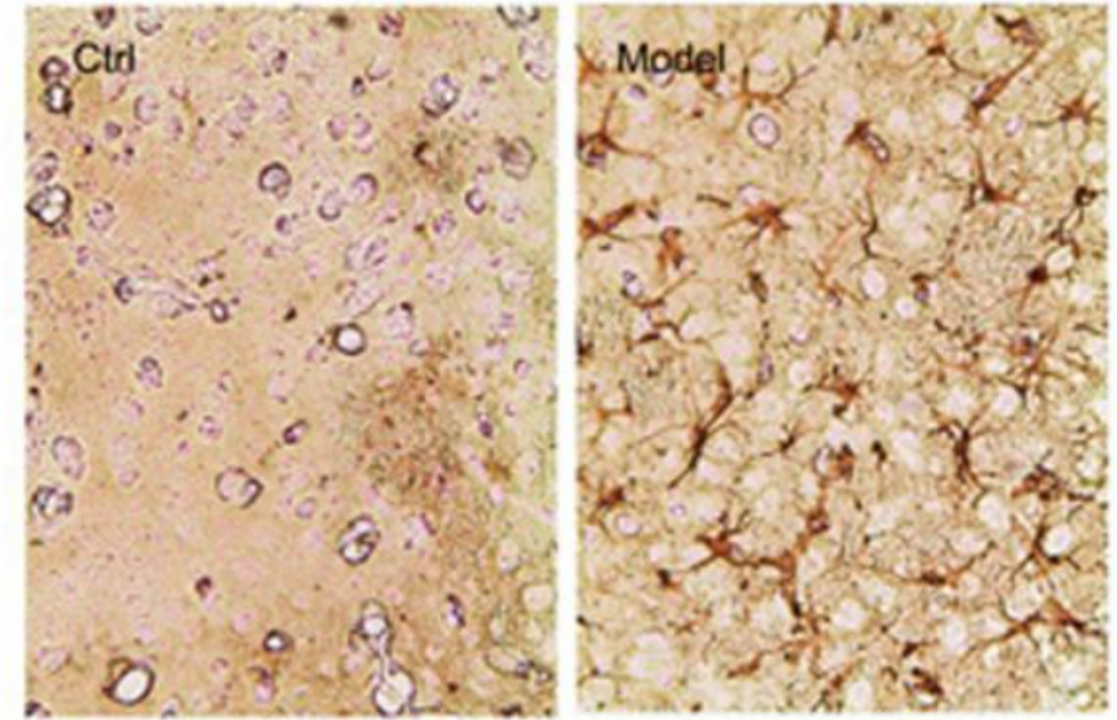
Figure 3. Accumulation of α-Syn[6]
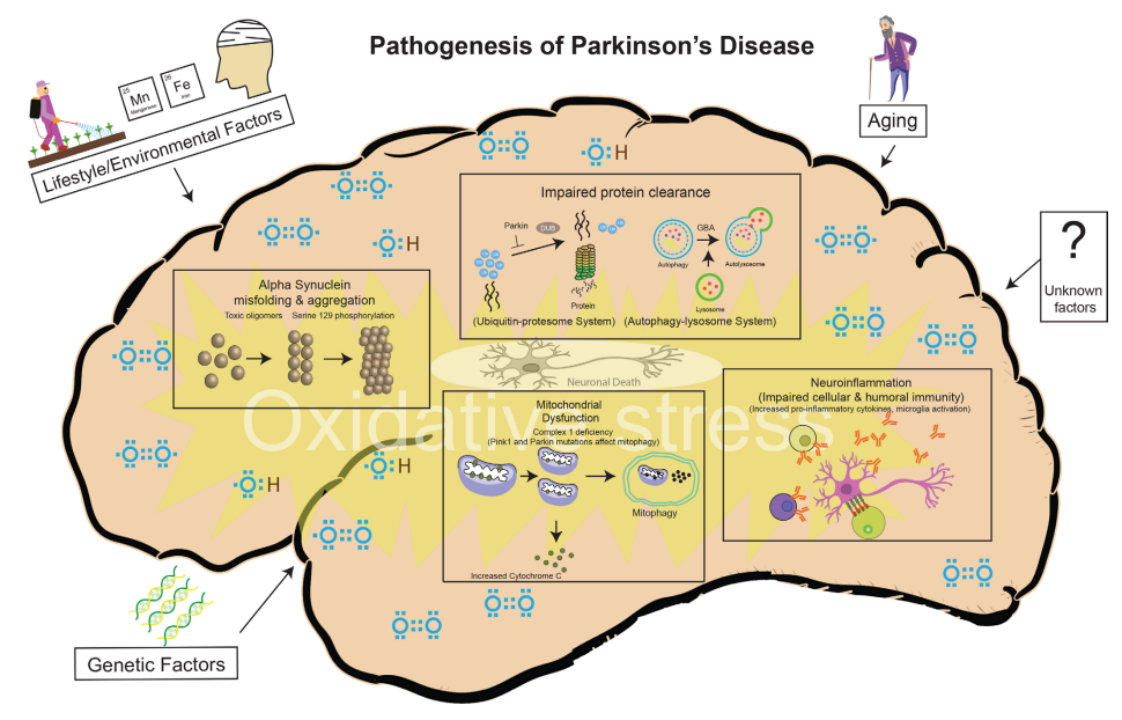
Figure 4. Risk Factors and Pathogenic Mechanisms of PD[4]
In recent years, researchers have gained a deeper understanding of the neurobiological mechanisms, genetic, and environmental factors influencing the development of PD. However, the road to drug development is still full of obstacles, such as: the lack of available biomarkers, good drug targets, and limited understanding of causative factors all hinder the progress of therapeutic development. To overcome these difficulties, research using appropriate animal models continues to be an invaluable approach.
There are two main methods of making animal models for PD: neurotoxin models and genetic models, with a small number of studies using models developed with the combination neurotoxin and genetic methods. Among all of these, neurotoxin models are the most widely used in PD research publications (Figure 5).[7]
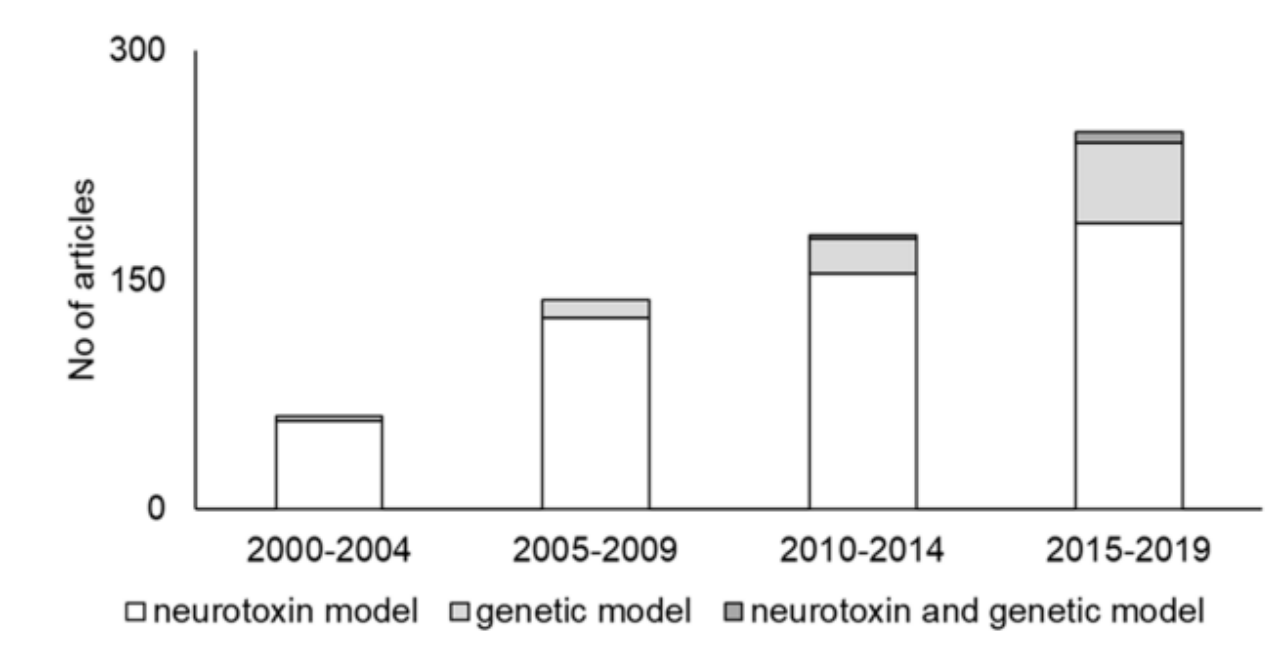
Figure 5. Trends in PD animal models. The number of articles gradually increased, while neurotoxin models remained the predominantly cited model.[7]
In neurotoxin models, 6-hydroxydopamine (6-OHDA) and 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) are the most widely used toxins. The methods for making neurotoxin models are mostly systemic injection and intracerebral injection of neurotoxin. Different neurotoxin models can simulate different pathological features of PD.
Modeling method: The injection sites are generally the striatum, medial forebrain bundle (MFB), and substantia nigra (SN). Apoptosis of DA neurons at the injection site can be detected after 3-5 days. 6-OHDA injection into the striatum destroys the axon terminals in the striatum followed by a slowly protracted retrograde degeneration of dopaminergic neurons in the SN. The symptoms are relatively mild and show a slow progression. However, 6-OHDA injection into the MFB and SN, which are also traditional targets, results in quick and massive dopaminergic cell degeneration.
Phenotypic features: This PD model is consistent with the clinical phenotype in terms of behavioral disturbances, while it develops the non-motor symptoms of PD but lacks the pathological phenotype of LB formation.
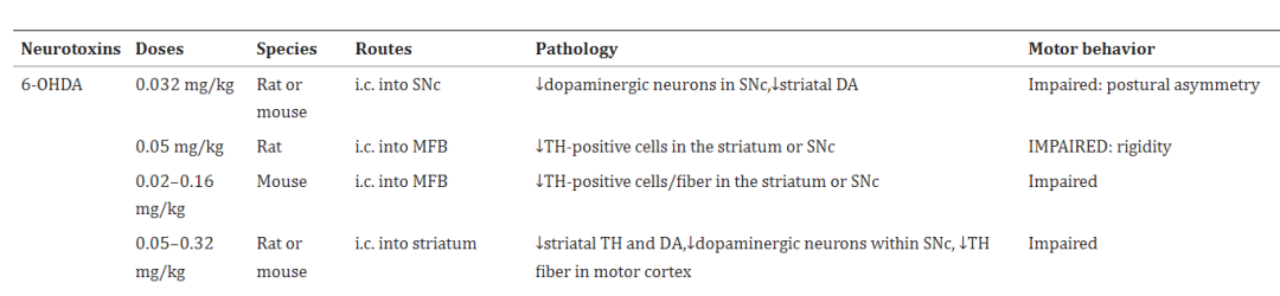
Figure 6. The Administration Routes of 6-OHDA and the resulting motor behavior changes (Parkinsonisms) in Rodent models[8]
Modeling method: MPTP is a lipophilic molecule that can easily cross the blood-brain barrier. After systemic or intracerebral injection, MPTP can be oxidized by monoamine oxidase B (MAOB) in astrocytes to the potent dopaminergic neurotoxin 1-methyl-4-phenylpyridinium ion (MPP+). MPP+ is a toxic metabolite that is readily taken up by dopaminergic neurons through the dopamine transporter (DAT) due to its structural similarity to dopamine.[9] Subsequently, MPP+ induced apoptosis of DA neurons in the SN occurs, accompanied by a decrease in striatal dopamine levels.[10] The mechanism of DA neuron apoptosis is that the ATP concentration in the striatum and SN decreases rapidly due to MPP+ inhibiting the respiratory chain complex I in mitochondrial respiration, and then DA neurons undergo apoptosis.[11]
Phenotypic features: Systemic injection of MPTP manifests in progressive pathological processes associated with PD, including loss of DA neurons in the striatum and SN, oxidative stress in the brain, and mitochondrial dysfunction. However, this model also lacks the pathological phenotype of LB formation.[8] In the existing neurotoxin models, the pathological phenotype of LB formation can be observed in the rotenone-induced PD model.[12]
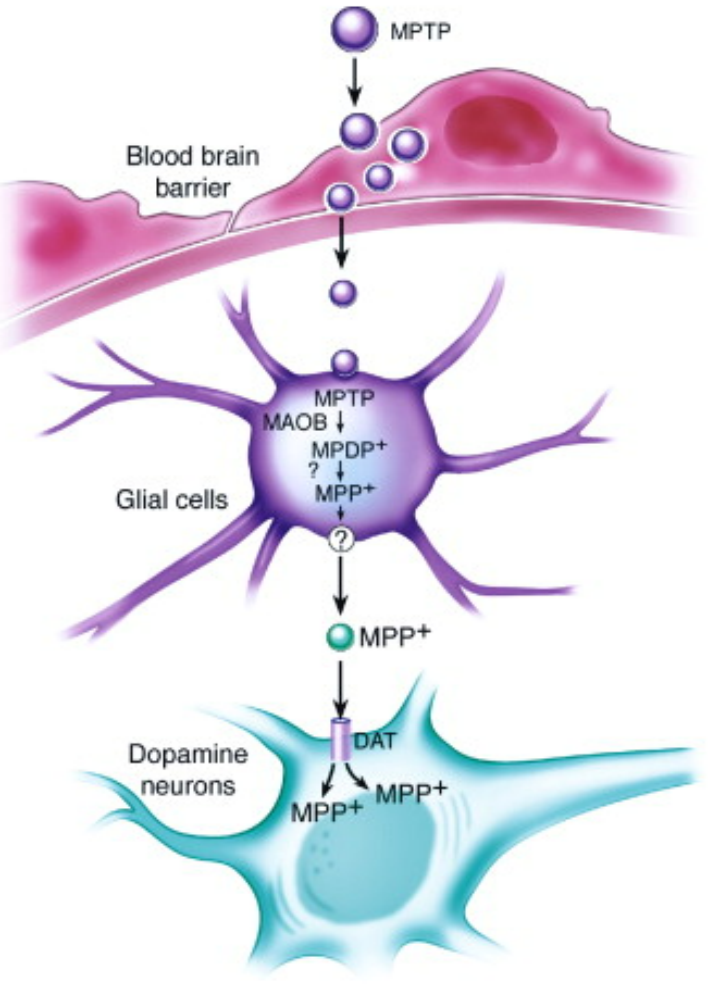
Figure 7. The mechanism of MPTP-induced apoptosis of DA neurons[5]
Genetics also plays an important role in the pathogenesis of PD. Through the analysis of familial PD and idiopathic PD, the following pathogenic genes were identified: SNCA, Parkin, DJ-1, PINK1, and LRRK2.[13] Information about these pathogenic genes and their related genetic models are shown in the table below:
|
Gene Name |
Gene Function |
Allele Modification Type |
Model Advantages |
Model Limitations |
|
SNCA |
Encodes α-Synuclein |
Mutations (A53T, A30P, and E46K) or repeating segments |
It can be used in research on α-Synuclein-related degeneration, and the relationship between genetic and environmental factors in PD. |
Some models have no significant loss of dopaminergic neurons, or exhibit patterns of apoptosis inconsistent with human PD pathology. |
|
LRRK2 |
Disease-causing gene of familial PD |
Mutations (G2019S and R1441C/G) |
It can be used in LRRK2 targeting drug testing and LRRK2 function research. |
This model exhibits motor deficits without significant loss of dopaminergic neuron degeneration and lack of LB formation. |
|
Parkin |
EncodesUbiquitin ligase; may lead to the accumulation of neurotoxic substrate after mutation or knockout |
Mutation, KO |
It can be used in Parkin function research. |
The model lacks phenotypic data. |
|
DJ-1 |
Encodesantioxidant protein |
KO |
It can be used with neurotoxin models. |
This model exhibits motor deficits, but lacks LB formation, no significant loss of dopaminergic neurons, and insufficient phenotypic data. |
|
PINK-1 |
Encodes Neuroprotective kinase |
Mutation, KO |
G309D-PINK1 mice exhibit a dopamine-decreased and motor-deficit phenotype. |
Most PINK-1 models do not show a reduction in dopaminergic neurons and dopamine levels. |
To accelerate your PD research, Cyagen provides researchers with KO/cKO mouse strains of PD-related genes. Click the links in the table below to view the corresponding product information and strain description!
|
Gene Name |
Tech Type |
Strain Name |
Product Details |
|
Snca |
KO |
C57BL/6N-Sncaem1Cya |
|
|
Snca |
cKO |
C57BL/6J-Sncaem1(flox)Cya |
|
|
Lrrk2 |
KO |
C57BL/6J-Lrrk2em1Cya |
|
|
Lrrk2 |
cKO |
C57BL/6J-Lrrk2em1(flox)Cya |
|
|
Park7 |
KO |
C57BL/6J-Park7em1Cya |
|
|
Park7 |
cKO |
C57BL/6J-Park7em1(flox)Cya |
|
|
Pink1 |
KO |
C57BL/6J-Pink1em1Cya |
|
|
Pink1 |
cKO |
C57BL/6N-Pink1em1(flox)Cya |
Looking for animal model supporting services? Cyagen’s neurobehavioral platform can provide rodent behavioral assessments, behavioral testing equipment, and professional recording and analysis systems, which includes: water maze, NORT, Y maze, rotarod, OFT, FST, EPM, and other types of rat and mouse behavioral testing services.
Please contact us so that our Model Design Specialists may promptly assist with your project needs. Alternatively, you can always email service-apac@cyagen.com to inquire about our services or obtain a quote.
[1] “Michael J. Fox: ‘I’m 61 years old, and I’m feeling it a little bit more’”. Andrea Jimenez. Elpais. 28 Oct 2022.
[2] Poewe W, Seppi K, Tanner CM, Halliday GM, Brundin P, Volkmann J, Schrag AE, Lang AE. Parkinson disease. Nat Rev Dis Primers. 2017 Mar 23;3:17013. doi: 10.1038/nrdp.2017.13. PMID: 28332488.
[3] World Health Organization, https://www.who.int/newroom/fact-sheets/detail/parkinson-disease.
[4] Jankovic J, Tan EK. Parkinson's disease: etiopathogenesis and treatment. J Neurol Neurosurg Psychiatry. 2020 Aug;91(8):795-808. doi: 10.1136/jnnp-2019-322338. Epub 2020 Jun 23. PMID: 32576618.
[5] Dauer W, Przedborski S. Parkinson's disease: mechanisms and models. Neuron. 2003 Sep 11;39(6):889-909. doi: 10.1016/s0896-6273(03)00568-3. PMID: 12971891.
[6] Zhang QS, Heng Y, Mou Z, Huang JY, Yuan YH, Chen NH. Reassessment of subacute MPTP-treated mice as animal model of Parkinson's disease. Acta Pharmacol Sin. 2017 Oct;38(10):1317-1328. doi: 10.1038/aps.2017.49. Epub 2017 Jun 26. PMID: 28649132; PMCID: PMC5630672.
[7] Kin K, Yasuhara T, Kameda M, Date I. Animal Models for Parkinson's Disease Research: Trends in the 2000s. Int J Mol Sci. 2019;20(21):5402. Published 2019 Oct 30. doi:10.3390/ijms20215402
[8] Zeng XS, Geng WS, Jia JJ. Neurotoxin-Induced Animal Models of Parkinson Disease: Pathogenic Mechanism and Assessment. ASN Neuro. 2018 Jan-Dec;10:1759091418777438. doi: 10.1177/1759091418777438. PMID: 29809058; PMCID: PMC5977437.
[9] Marti Y., Matthaeus F., Lau T., Schloss P. Methyl-4-phenylpyridinium (MPP+) differentially affects monoamine release and re-uptake in murine embryonic stem cell-derived dopaminergic and serotonergic neurons. Mol. Cell. Neurosci. 2017;83:37–45. doi: 10.1016/j.mcn.2017.06.009.
[10] Bezard E., Gross C.E., Fournier M.C., Dovero S., Bloch B., Jaber M. Absence of MPTP-induced neuronal death in mice lacking the dopamine transporter. Exp. Neurol. 1999;155:268–273. doi: 10.1006/exnr.1998.6995.
[11] Sayre L.M., Wang F., Hoppel C.L. Tetraphenylborate potentiates the respiratory inhibition by the dopaminergic neurotoxin MPP+ in both electron transport particles and intact mitochondria. Biochem. Biophys. Res. Commun. 1989;161:809–818. doi: 10.1016/0006-291X(89)92672-7.
[12] Inden M., Kitamura Y., Abe M., Tamaki A., Takata K., Taniguchi T. Parkinsonian rotenone mouse model: Reevaluation of long-term administration of rotenone in C57BL/6 mice. Biol. Pharm. Bull. 2011;34:92–96. doi: 10.1248/bpb.34.92.
[13] Chia SJ, Tan EK, Chao YX. Historical Perspective: Models of Parkinson's Disease. Int J Mol Sci. 2020 Apr 2;21(7):2464. doi: 10.3390/ijms21072464. PMID: 32252301; PMCID: PMC7177377.
We will respond to you in 1-2 business days.